Blister Packaging: Reduce Overdose
According to Wikipedia, "Opioids are substances that act on opioid receptors to produce morphine-like effects. Medically they are primarily used for pain relief, including anesthesia." Opioids have a significant tendency to have side effects, and such drug itself has serious problems of substance abuse and addiction.
Only 5% of the world's population of Americans consume more than 80% of the world's opioids. According to statistics from the US Department of Health and Human Services, the opioid crisis has had a profound impact on the United States, resulting in 130 deaths per day. In addition, according to a data analysis by the National Safety Council, the odds of accidental death among Americans due to overdose of opioids are higher than the chance of dying from a car accident. Therefore, the misuse and abuse of opioids remains one of the priorities that the FDA needs to address.
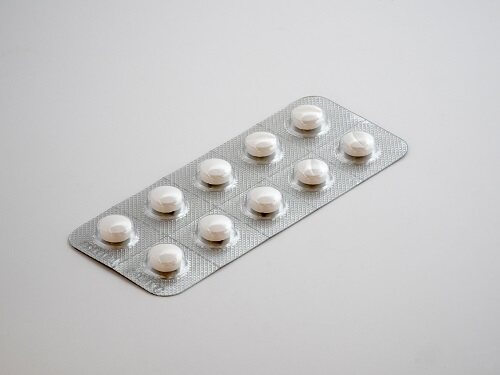
The ideal state of opioid use is to find a balance point to ensure that such substances are used only in medical and scientific research without being abused. On May 30, the US Food and Drug Administration's proposal called for the use of blister packs for some opioids. It aims to enable prescriptions to make more reasonable prescriptions based on clinical needs by changing the packaging of the drug.
To a certain extent, limiting the packaging of pharmaceuticals and the use of unit dose packages may reduce overdose and lethality. The convenience of such packaging can encourage the prescriber to choose a shorter period of use, thereby limiting the amount of opioid drug that is dispensed to the patient. If the blister pack dose of the drug is consistent with the time of administration, it helps to reduce the overall drug distribution. Unit dose packaging can reduce oversupply of market products caused by poor prescriptions. In addition, it is easier to track than bottled pills, helping to help patients avoid accidental overdose.
Leave your comment
Also Offers


Containment Automatic Capsule Filling Machine SFK-703

Fully Automatic Dosator Capsule Filling Machine CZ-40

Our Team
As an expert in the pharmaceutical and pharmaceutical packaging industry, iPharMachine has provided solutions for hundreds of pharmaceutical and health product manufacturers for 17 years. By visiting customers, we get good reviews from our customers.
- info@ipharmachine.com
- English Español Deutsche







