Pharmaceutical Tablets Packaging: Aluminum Plastic Strip Composite Film
For some larger dosage forms of pharmaceutical tablets or pharmaceutical tablets that are sensitive to ultraviolet light, strong in hygroscopicity, or that require heat and cold resistance, long effective period or improve packaging grade, most of them are strip-shaped composite film packaging, referred to as SP. (strip Packaging).
It uses two layers of medicinal strip packaging film (SP film) to sandwich the tablets, and the unit medicines are separated by a certain distance. The two layers of SP film around the medicine are heat-sealed on the strip packaging machine. The heat-sealing between pills forms in a unit package form (single piece package or in a row to form a small package).
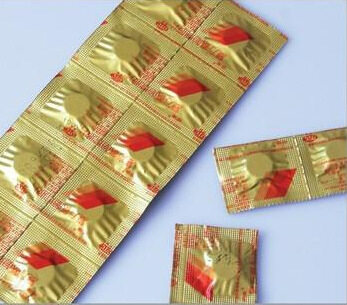
The strip packaging is continuous operation on the strip packaging machine, which is especially suitable for large-volume automatic packaging. When taking the medicine, the SP film can be torn along the tooth marks. The strip packaging film has a certain tensile strength and elongation, which is suitable for medicines of various shapes and sizes, and closely attached to the medicine after packaging, and not easy to cause cracks and wrinkles.
Up to now, aluminum-plastic composite film is commonly used, such as cellophane and aluminum foil and polyethylene composite (PT/AL/PE), polyester and aluminum foil and polyethylene composite (PET/AL/PE), that is,the aluminum foil and the plastic film are laminated or extruded by an adhesive. The composite film is composed of a base layer, a printing layer, a high barrier layer and a sealing layer. The base layer is outside, the heat-sealing layer is included, and the high barrier layer and the printing layer are located in the middle. The base material is required to have excellent mechanical properties, good gloss printability, good transparency, high barrier property, safety, non-toxicity, and no heat sealability. Typical materials are polyester (PET), cellophane (PT) and cellophane with PVDC coating. The high barrier layer needs to have good gas barrier properties and moisture resistance, good mechanical properties, a certain elongation, is resistant to cold and heat, and be safe and non-toxic. The typical material is soft aluminum foil. However, this soft aluminum foil is opaque, does not rust itself, and has strong opacity. If a transparent strip packaging film is required, PVDC should be used as a high barrier material. PVDC is a high barrier material, and its biggest feature is its excellent barrier properties to gas and water vapor. The sealing layer is an inner layer of a strip-shaped packaging film, which has excellent heat-sealing properties, chemical stability and safety and hygienic properties, and generally uses a low-density polyethylene material.
The choice of pharmaceutical packaging for pharmaceutical packaging is determined by the nature of the drug (moisture, oxidation, retention of medicinal properties, etc.) and shelf life. It mainly based on the quality characteristics of the drug requirements, combined with the characteristics of the composite product. Several medicinal composite membrane features are provided below for reference:
- a. Ordinary composite film: Composite structure with polyester and aluminum foil and polyethylene composite (PET / AL / PE), characterized by good printing adaptability, good gas and moisture barrier properties for drugs.
- b. Medicinal strip-like easy-to-tear composite film: Composite structure made of cellophane and polyethylene and aluminum foil (PT/PE/AL/PE). It has good tearability and is convenient for consumers to access the product. Good gas and water vapor barrier properties ensure a long shelf life of the contents. It is suitable for the packaging of effervescent agents, paints, tablets and capsules.
- c. Paper aluminum-plastic composite film: The composite structure is made of paper and polyethylene and aluminum foil (paper/PE/AL/PE), characterized by good printability and good stiffness. It has good barrier properties to gas or water and can guarantee a long shelf life of the drug.
Knowing another packaging - blister packaging, click here.
Leave your comment
Also Offers


Containment Automatic Capsule Filling Machine SFK-703

Fully Automatic Dosator Capsule Filling Machine CZ-40

Our Team
As an expert in the pharmaceutical and pharmaceutical packaging industry, iPharMachine has provided solutions for hundreds of pharmaceutical and health product manufacturers for 17 years. By visiting customers, we get good reviews from our customers.
- info@ipharmachine.com
- English Español Deutsche







